Quality Management System
Based on ISO 22716/ ISO 9001 and CGMP certification facilities,
Amaranth OEM / ODM systems help vendors and distributors maximize their competitive edge in the market with the highest quality,
reasonable price and timely supply. Professional researchers and field engineers understand exactly what customers want,
and quickly follow the entire process from planning to production.
Quality objectives
-
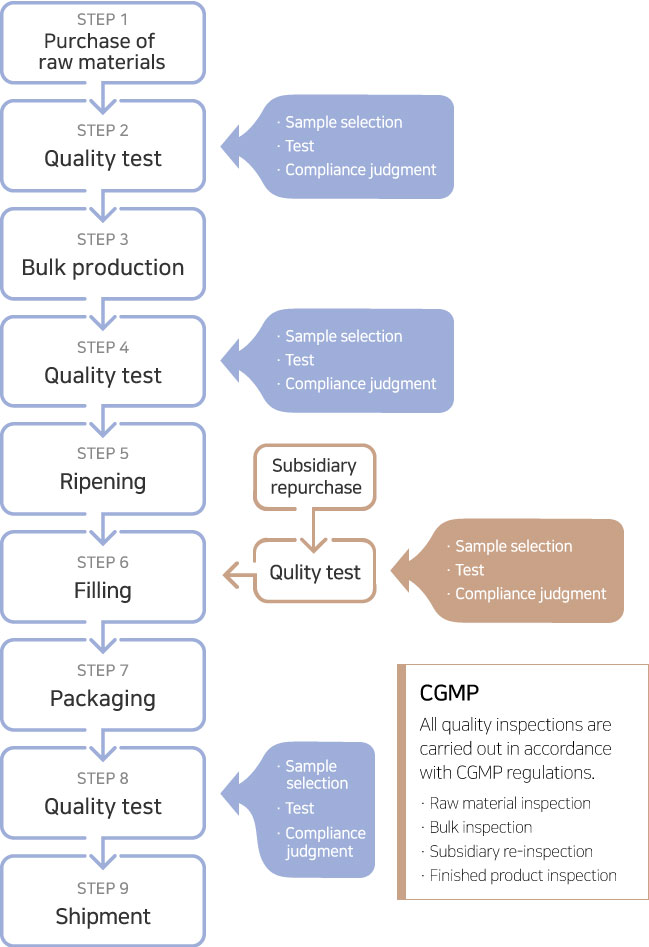
STEP 1
Purchase of raw materials
Accurately understand customer needs for quality
-
STEP 2
Quality test
Accurate production and delivery compliance
-
STEP 3
Bulk production
Visual inspection and a thorough inspection system for specifications and physical properties are established to prevent claims that occur after shipment.
-
STEP 4
Quality test
Cost reduction is realized by minimizing the defect rate (100 ppm) and improving productivity by developing and maintaining an optimal production process.
Process
Production process
Inertia test
In the raw material inspection, sensory and visual inspections such as color, odor,
and feeling are performed, and qualitative inspections such as component content and
viscosity of raw materials are also conducted using various test equipment.
Inspection of packaging materials includes pumping tests,
inflow and resistance tests, and peeling tests for size and color products for the fastening state,
appearance and surface of the jointed part.
This inspection is to thoroughly check the raw material/packaging material by re-inspecting even
the verified raw material/packaging material.
Raw material weighing
In order to manufacture the contents,
it is necessary to measure the raw materials according to the characteristics of each product.
Content manufacturing
After the measurement has been completed, the transferee checks the raw materials once more,
and then puts the raw materials into the manufacturing facility tank for manufacturing.
The manufacturing of these cosmetics is controlled by the manufacturing management system,
and through this, excellent products are produced.
Content filling
The supplied container is subjected to visual or air work according to
its characteristics to remove the remaining foreign substances, and the contents filling operation begins.
Microbial testing
Microbial inspection is the basis of thorough product quality control,
and it manages not only raw materials/products,
but also air microorganisms, dropping bacteria,
and surface bacteria in the manufacturing/packaging room,
so that customer trust and problems that may occur during customer use are predicted/removed.
Microbial testing is carried out in the following steps,
and through these, only suitable samples (raw materials/contents/products) are released.
※ Sample reception > Experiment preparation > Microbial test > Culture > Judgment > Result input
Shipment
Only products that meet all quality tests such as sensory tests,
analytical tests, and microbiological tests are shipped.